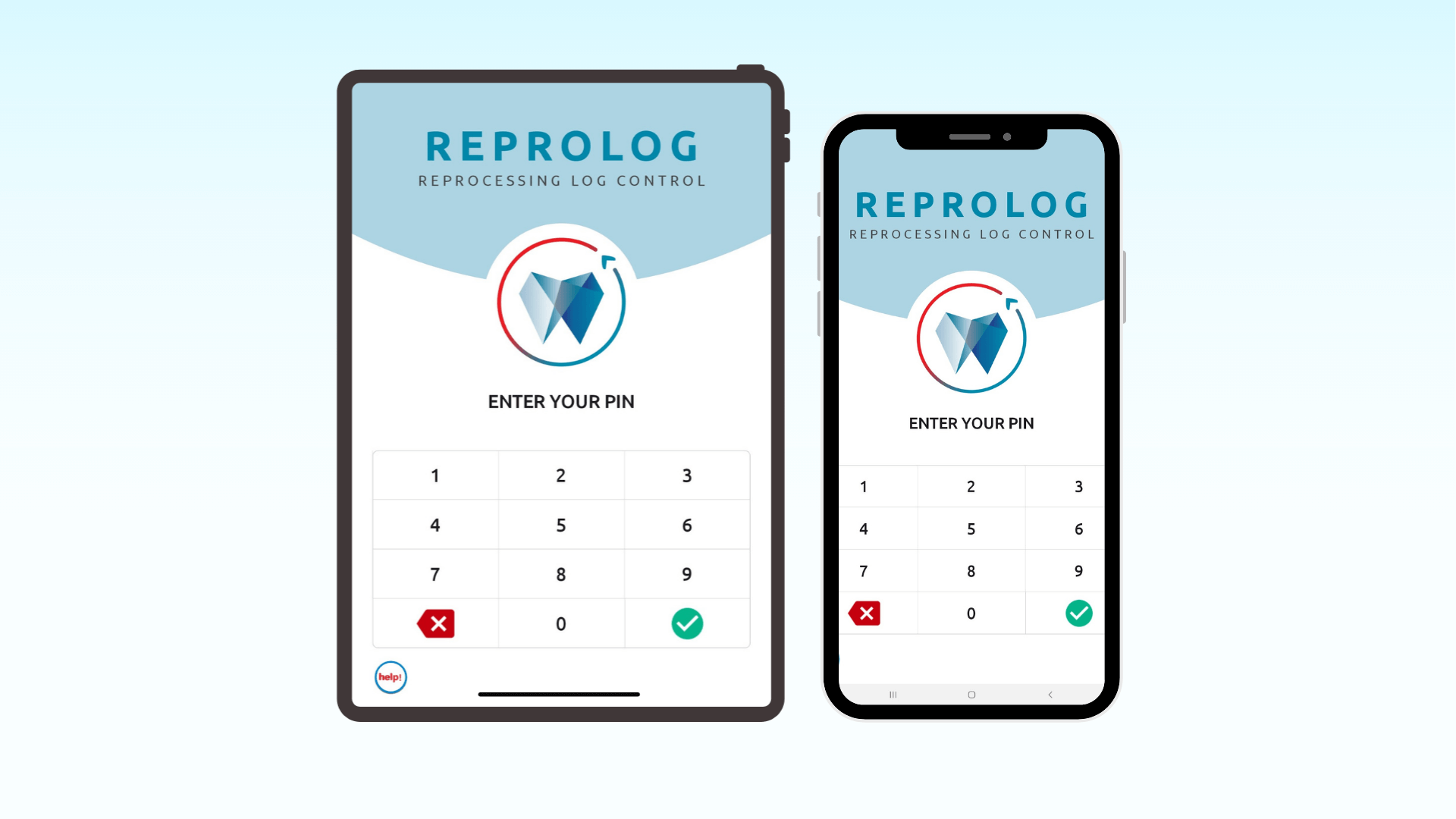
How Reprolog Solves
Common IPAC Lapses
in Dental Clinics
- February 09, 2026
Infection Prevention and Control (IPAC) compliance is a critical responsibility for dental clinics. Despite established protocols, gaps in sterilization practices and documentation remain widespread, often caused by human error, inconsistent workflows, or lack of real-time oversight. These lapses can compromise patient safety, trigger regulatory non-compliance, and create operational inefficiencies.
Below, we break down common IPAC lapses and demonstrate how Reprolog systematically resolves them across three core areas.
Establishing a Compliant Workflow
Improper instrument flow is a major contributor to IPAC lapses. Maintaining a strict one-way flow from contaminated to sterile areas is essential to prevent cross-contamination, yet many clinics struggle to enforce this consistently.
IPAC Lapses:
Reprocessing areas lack a one-way workflow, increasing cross-contamination risk
Sterile and non-sterile items are stored or handled improperly
Inadequate sterilization processes, including inconsistent quality assurance and improper instrument handling
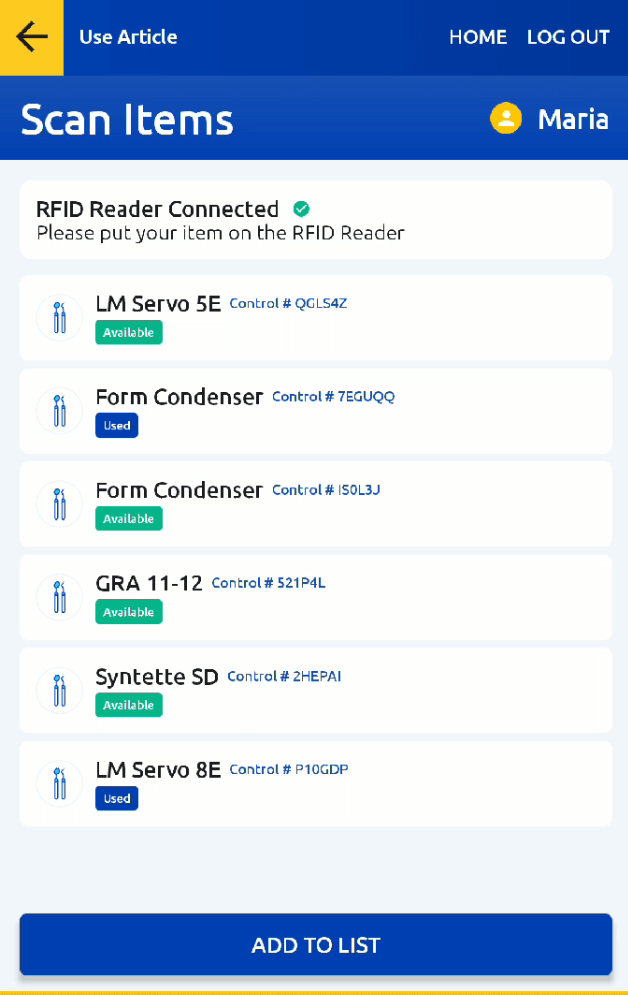
Solution: Reprolog enforces a dirty-to-clean workflow, tracking every instrument through its full lifecycle—from use, through reprocessing and sterilization, back to point-of-use. The system validates sterilizer cycles, including BI and CI results, and prevents the release of instruments if packaging, sterilization, or QA requirements are unmet. Reprolog also tracks instrument use and return, linking each instrument to the patient it was used on, providing complete traceability and accountability.
Standardizing Best Practices
Even with proper workflows, lapses in sterilization quality assurance and compliance can occur if best practices aren’t consistently applied. Standardizing procedures ensures every sterilizer cycle, indicator check, and instrument handling step meets IPAC requirements.
IPAC Lapses:
Utilization of expired instruments or chemical indicators
Sterilization pouches lacking internal chemical indicators or proper labeling with lot/batch numbers
Failure to conduct biological testing of sterilizers at least once every two weeks
Inconsistent or incomplete sterilizer quality assurance, increasing the risk of ineffective sterilization
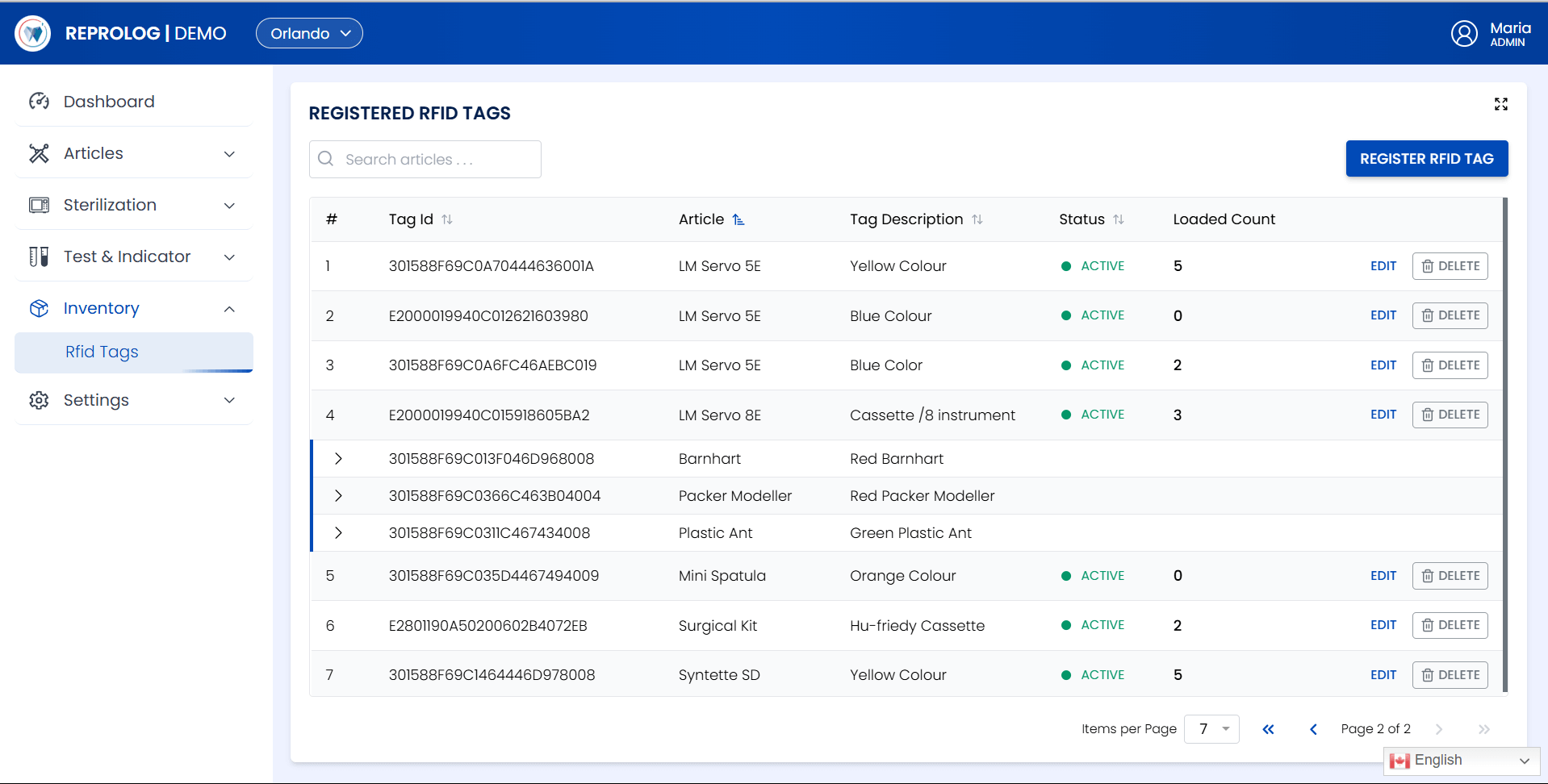
Solution: Reprolog has best practices built directly into the system, standardizing every sterilization workflow to automate compliance and eliminate human error. For example, before sterilizing an implantable device, the system notifies the user that a biological indicator (BI) is required, and the instrument cannot be used until the BI validation is passed. Expired instruments are automatically flagged and tracked, preventing use until they are properly re-sterilized. All sterilization pouches are monitored to ensure correct internal indicators and labeling. Testing cycles are enforced automatically—BI testing for all autoclaves is scheduled and tracked at least once per week—ensuring consistent adherence to IPAC standards.
Audit Readiness at All Times
Regulatory inspections demand accurate, accessible, and complete records. Manual record-keeping often results in missing load details, lot numbers, or maintenance logs, putting clinics at risk of non-compliance.
IPAC Lapses:
-
Incomplete or missing sterilizer load documentation
-
Lot numbers and expiry dates of single-use devices not recorded
-
Failure to document load contents or commercially purchased pre-packaged sterile items